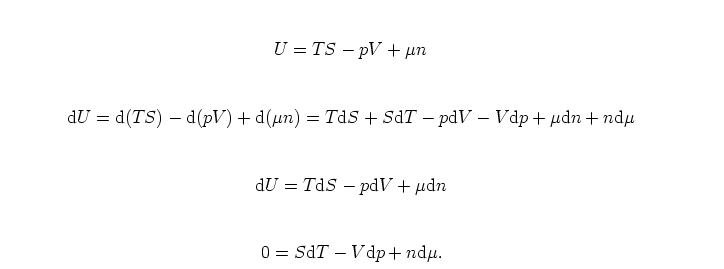Intensive Functions and Extensive FunctionsThermodynamics Variables are either extensive or intensive. To illustrate the difference between these kings of variables, think of mass and density. The mass of an object depends on the amount of material in the object, the density does not. Mass is an extensive variable, density is an intensive variable. In thermodynamics, T, p, and are intensive, the other variables that we have met, U, S , V , N, H, F, and G are extensive. We can develop some useful formal relationships between thermodynamic variables by relating these elementary properties of thermodynamic variables to the theory of homogeneous functions.
The energy form of the Euler equation
This is the Gibbs-Duhem equation. It shows that three intensive variables are not independent - if we know two of them, the value of the third can be determined from the Gibbs-Duhem equation.