|
In our surroundings there are mixtures of two or more components. Those mixtures are either homogeneous or heterogeneous. Homogeneous mixtures are uniform in composition while heterogeneous mixtures are not uniform in composition.
For example, Air is a homogeneous mixture and oil in water is a heterogeneous mixture. Homogeneous and heterogeneous mixtures can be separated into their components by several physical methods. The choice of separation techniques is based on the type of mixture and difference in the chemical properties of the constituents of a mixture. |
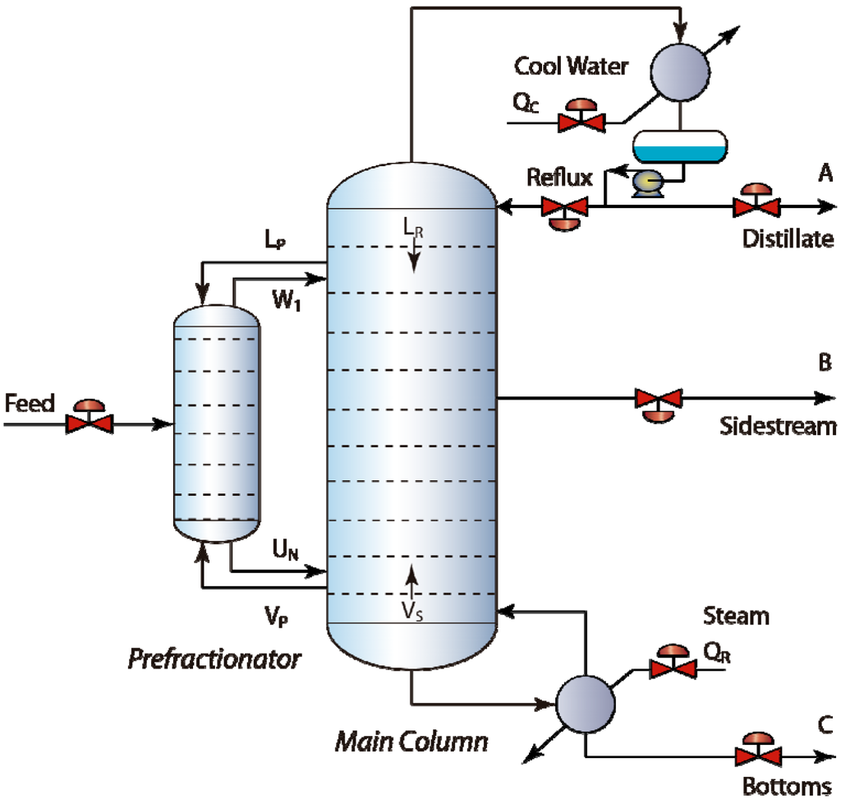
Types OF SEPARATION PROCESSES
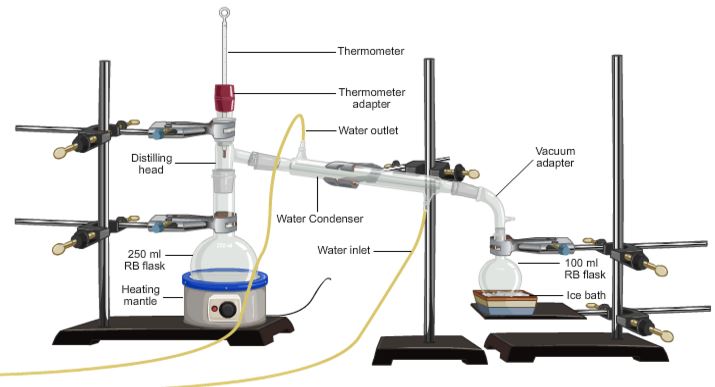
SIMPLE DISTILLATION Is a method used for the separation of components of a mixture containing two miscible liquids that boil without decomposition and have sufficient difference in their boiling points.
|
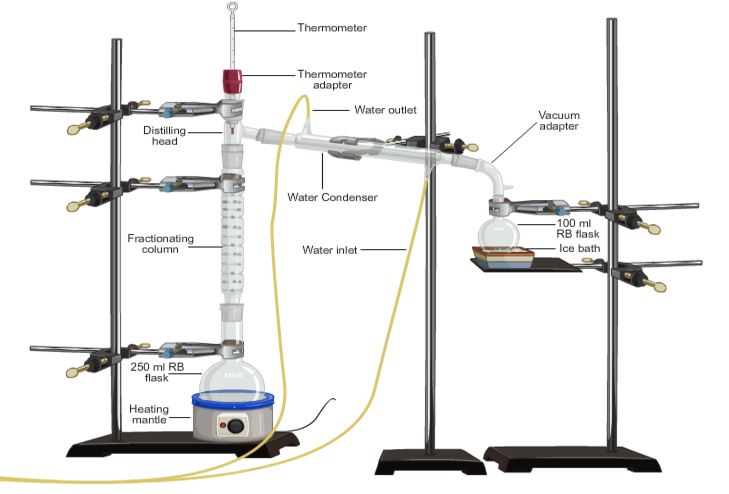
fractional distillationFractional distillation is used for the separation of a mixture of two or more miscible liquids for which the difference in boiling points is less than 25K.
|
azeotropic distillationIs the specific technique of adding another component to a mixture in order to generate a new lower-boiling mixture to break an azeotrope in distillation.
|
Raoult´s law
Raoult's law states that the vapor pressure of a solvent above a solution is equal to the vapor pressure of the pure solvent at the same temperature scaled by the mole fraction of the solvent present.
YAP=XAP*
Where:
YA: gas composition
P: system pressure
XA: Liquid composition
P*: Antoine law
YAP=XAP*
Where:
YA: gas composition
P: system pressure
XA: Liquid composition
P*: Antoine law
Antoine Law
Felder, Richard M. Rosseau, Ronald W.. (2005). Elementary Principles of Chemical processes. México: Limusa Wiley. (pag. 640-641)