Phase Diagram in Thermodynamics
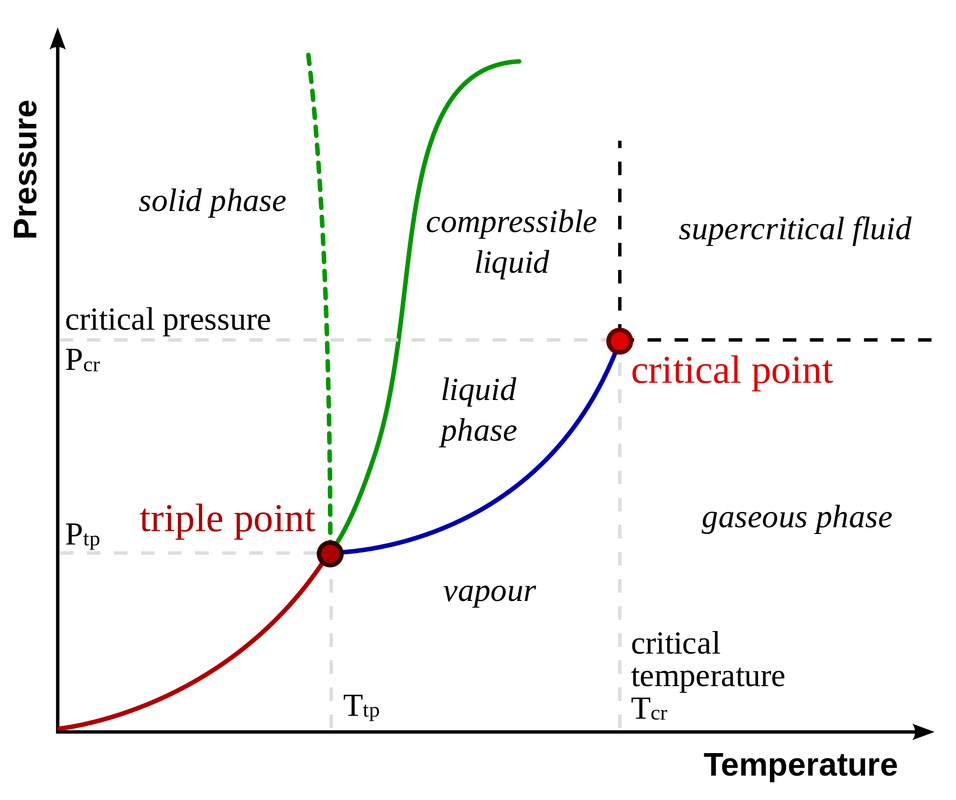
Phase diagram is a graphical representation of the physical states of a substance under different conditions of temperature and pressure. |
A typical phase diagram has pressure on the y-axis and temperature on the x-axis. As we cross the lines or curves on the phase diagram, a phase change occurs. In addition, two states of the substance coexist in equilibrium on the lines or curves.
|
Phase diagram (Graph)
What's what?
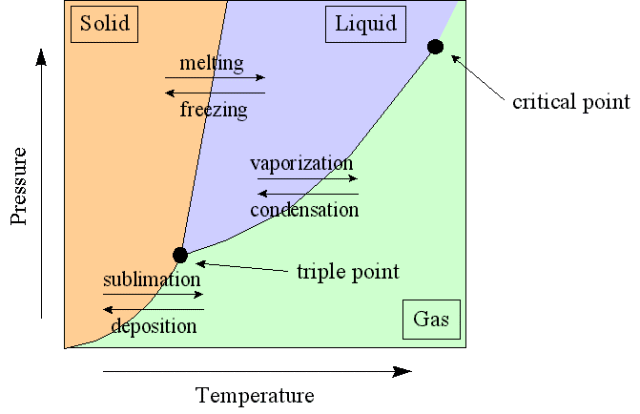
- Triple point – the point on a phase diagram at which the three states of matter: gas, liquid, and solid coexist
- Critical point – the point on a phase diagram at which the substance is indistinguishable between liquid and gaseous states.
- Fusion(melting) (or freezing) curve – the curve on a phase diagram which represents the transition between liquid and solid states.
- Vaporization (or condensation) curve – the curve on a phase diagram which represents the transition between gaseous and liquid states.
- Sublimation (or deposition) curve – the curve on a phase diagram which represents the transition between gaseous and solid states.
It's very important to have in mind that each pure component has it's own diagram! Because it depends on it's physical and chemical characteristics!
We add some examples down here:
Talking about the new changes that a system suffer when it changes it's phases, we have the following phenomenoms:
- Sublimation is when the substance goes directly from solid to the gas state.
- Deposition occurs when a substance goes from a gas state to a solid state; it is the reverse process of sublimation.
- Melting occurs when a substance goes from a solid to a liquid state.
- Fusion is when a substance goes from a liquid to a solid state, the reverse of melting.
- Vaporization is when a substance goes from a liquid to a gaseous state.
- Condensation occurs when a substance goes from a gaseous to a liquid state, the reverse of vaporization.
- Solid-Liquid boundary: dp/dt = delta H of fussion/ T Delta volume of fussion
- Liquid-vapour boundary: dp/dt = delta H of vaporization/ T Delta volume of vaporization
- Solid - vapour: dp/dt = delta delta H of sublimation/ t Delta volume of sublimination