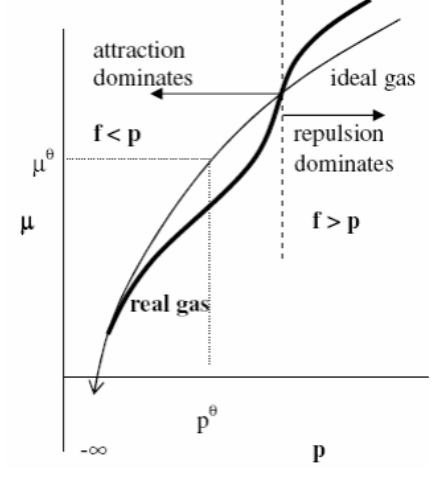
Fugacity
AN iDEAL GAS is a gas that conforms in physical behavior, an idealized relation between pressure, volume and Temperature. |
Fugacity is the thermodynamic drive for molecules to escape from one phase to another.
Chemicals diffuse from high to low fugacity, just as heat travels from high to low temperature. When a chemical's fugacity in one phase equals the fugacity in another, it is said that they are in equilibrium.
|
References:
KING ABDULAZIZ UNIVERSITY, (2015). Fugacity. [online] Available at: http://www.kau.edu.sa/Files/0053615/files/16538_Lecture%207%20phase%20equilibrium.pdf
KING ABDULAZIZ UNIVERSITY, (2015). Fugacity. [online] Available at: http://www.kau.edu.sa/Files/0053615/files/16538_Lecture%207%20phase%20equilibrium.pdf